Calculation of K eq and Concentrations: After you have finished this you should be able to: 1) Calculate the equilibrium constant. 2) Calculate the equilibrium concentration of a participant when the value of K eq is known as well as the concentrations of the other participants. 3) Determination of the net direction of a reaction prior to establishing an equilibrium. Chemistry Stack Exchange is a question and answer site for scientists, academics, teachers, and students in the field of chemistry. It only takes a minute to sign up.
- How To Calculate Keq Of A Reaction
- How To Calculate Keq From Concentrations
- How To Calculate Keq Thermo
- How To Calculate Keq From Delta G
- How To Calculate Keq With Temperature
Great Thanks to Tina Nye (A-5; 05-06) for much work on graphics: making a rough idea reality!
Chemical equilibrium
A system in equilibrium is like our ants up there! As long as the ants work at the same speed, the piles of sand remain in equilibrium. Neither gets bigger or smaller (It is important to note however that the piles are not the same size!). The work that each ant does exactly offsets what the other is doing. Products are being stacked up and the same rate that they are being taken away. Reactants are being stacked up at exactly the same rate that they are being taken away. If this were a chemical reaction, reactants would be colliding to make product at the same rate that products would be colliding with each other to make fresh reactant again! The reaction never really stops, but the forward reaction and reverse reaction proceed and the same rate, so we don't see a change in the size of the piles! Nifty Huh!!!!?
Chemical equilibrium is reached when the rates for the forward and reverse chemical reactions are equal for a chemical system. Or you could say when products are being made as fast as they are breaking down to form reactants again. The concentrations of products and reactants are generally NOT EQUAL.
To know what the concentrations are, you use the Equilibrium Constant expression (Keq).
Nba 2k17 crack online. Keq = [products]/[reactants]. Its a ratio : ) So Keq > 1 favors products, Keq < 1 favor reactants.
For a general equation like:
Sketchup 2017 download mac. wA + xB <> yC + zD How to install msr605 for mac.
Remember, the square brackets mean concentration, in molarity, if it is given in some other unit you must calculate molarity. Coefficients in the chemical equation become exponents in the Keq expression. Leave out any solids or pure liquids since they have undefined molarities ; ).
Mlb 2k12 pc keygen no virus download. Keq= If you spend time with the following notes, you'll master this topic in no time.
Predicting final concentrations and cheating while were doing it.
How To Calculate Keq Of A Reaction
Equilibrium Constants
Equilibrium is a state of balance between products and reactants. When the rate of the forward reaction is equal to the rate of the reverse reaction, the concentrations of products and reactants remain constant. Every reaction is striving to reach equilibrium.
Le Chatelier’s Principle : if equilibrium is disturbed (eg. if product or reactant concentrations are altered), the direction of the reaction will shift to restore the state of equilibrium.
Equilibria are reflections of Le Chatelier’s Principle. If we begin a reaction with only the reactants, they will readily form products. As the concentration of the products increases, the rate of the reverse reaction increases. As the concentration of reactants decreases, the rate of the forward reaction decreases. Eventually the reaction rates will cancel each other out, and there will be no net change in the concentration of reagents.
The Equilibrium Constant(Keq) describes the ratio of products to reactants in a state of equilibrium.
- Some reactions have a big Keq. When these reactions reach equilibrium, there will be a lot of product, and little reactant left.
- Some reactions have a small Keq. Don’t expect these reactions to produce much product.
- See that the Keq for the reverse reaction will be the reciprocal of the forward reaction.
Strong acids and bases dissociate with large equilibrium constants.
We use a specific type of equilibrium constant when describing the dissociation reactions of acids and bases. Recall the equation for the dissociation of an acid:
The Keq of this reaction is given by. We call it Ka, the Acid Dissociation Constant
Likewise, the equilibrium constant for a base dissociation is given by Kb, its Base Dissociation Constant.
The Dissociation Constant determines the strength of the Acid or Base
Strong vs weak Acids
Here is a list of many acids along with their Ka values. The top three on the list would be considered strong acids – they will essentially dissociate completely. Most acids are weak, though two weak acids can vary greatly in Ka.
Most important difference between strong and weak acids: once the reaction reaches equilibrium, strong acids will be dominantly in the conjugate base form. Weak acids will exist as a mixture of the original acid and the conjugate base form.
For simplicity, the strength of the acid will often be given as pKa. pKa represents the same information as Ka except its logarithmic scale eliminates the need for exponential notation. This simple equation converts between Ka and pKa.
Using these equations, see how a large pKa denotes a very weak acid. Bases have very large pKa
How To Calculate Keq From Concentrations
Bases use Kb and pKb as measurement of how favorably they accept protons (basicity). Kb works exactly like ka. A small Kb means equilibrium highly favors conjugate acid formation, indicating a strong base. A larger Kb indicates a weak base that will partially form conjugate acid and partially remain in its base form.
General Note: A stronger acid or base will produce a weaker conjugate. A weaker acid or base will produce a stronger conjugate.
Summary
Here is an example of an acid base reaction. The Keq is 1.8e-5
Do we start with an acid or a base?
NH3 (ammonia) is a base. This is clear because the products include its protonated form NH4 (ammonium). We see it is acting like a base and accepting a proton from water.
The conjugate acid is ammonium. In the reverse reaction, ammonium acts as an acid and hydroxide acts as a base. Ammonium donates a proton to hydroxide.
Find Kb for ammonia: This would simply be the given Keq for the reaction. Kb is just the Keq of a specific type of reaction between a base and water. Kb = 1.8e-5. Ammonia is a weak base.
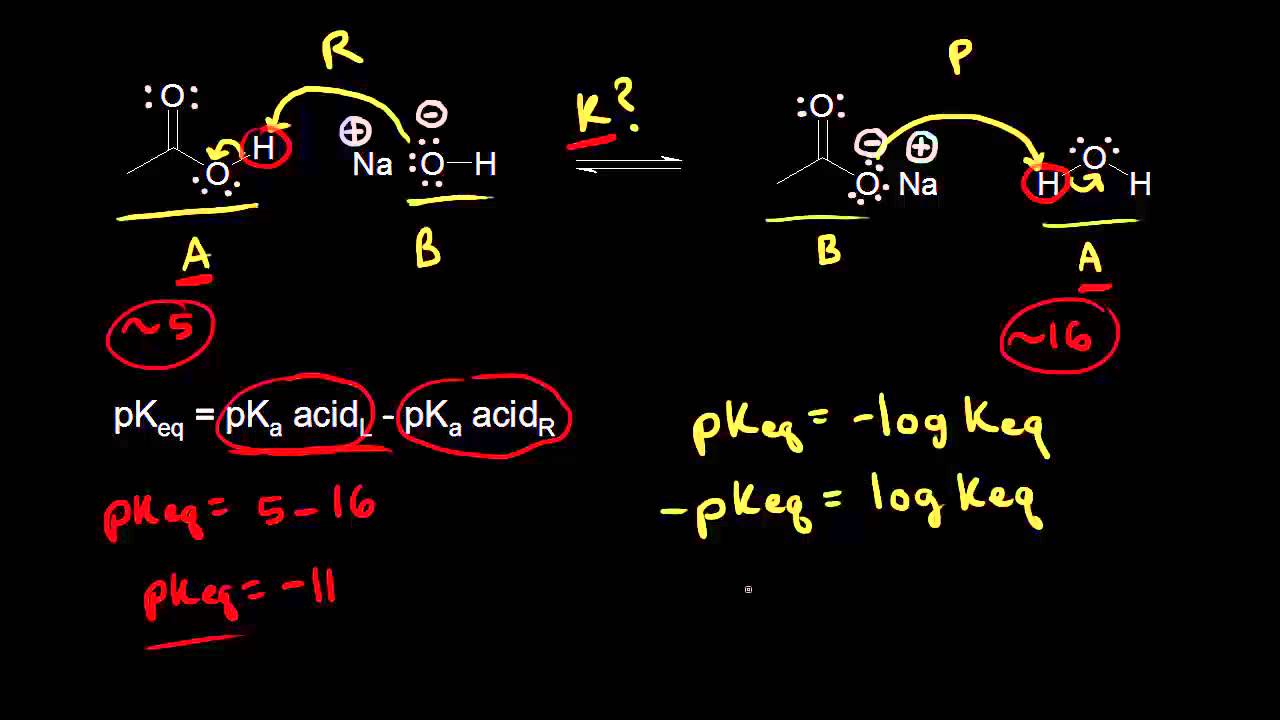
Find Ka of conjugate acid. This is would equal the Keq of the reverse reaction. The Keq of the reverse reaction is the reciprocal of the the forward reaction. Ka = 1/Kb = 5.6e-10. Since ammonia is a base of moderate strength (though defined as ‘weak’), see how its conjugate acid is is very very weak. This makes sense because if the conjugate acid were stronger, the reverse reaction would be more favorable, causing the forward base reaction to become less favorable.
Convert Ka and Kb to pKa and pKb.
pKb = 4.75 pKb = 9.25
How To Calculate Keq Thermo
Note: pKa + pKb = 14.0
How To Calculate Keq From Delta G
After reading this you should be able to:
How To Calculate Keq With Temperature
- make a prediction about the properties of a molecule by looking at its pKa value.
- label the acid, base, conjugate acid, and conjugate base in a reaction
- Use Keq to determine concentrations of reagents.

